Fluorphlogopite - synthetic mica
 Fluorphlogopite mica (also called artificial mica, synthetic mica crystal) belongs to artificial mica crystal of silicate. It is the mica crystal which is smelted in a platinum crucible at the condition of high temperature of 1500℃ and the condition of process and the material ratio is controlled strictly.
Fluorphlogopite mica (also called artificial mica, synthetic mica crystal) belongs to artificial mica crystal of silicate. It is the mica crystal which is smelted in a platinum crucible at the condition of high temperature of 1500℃ and the condition of process and the material ratio is controlled strictly.
The synthetic fluorine mica, fluorphlogopite KMg3(AlSi3O10)F2, contains no (OH)- of the natural phlogopite KMg3(AlSi3O10)(OH)2. The (OH)- is fully substituted with F- . Large fluorine mica crystals of high quality are grown by a Bridgman-Stockbarger method using platinum crucibles with seeds.
The synthetic mica's high temperature resistance, corrosion resistance and light transmission performance is beyond compare. It can work at the high temperature of 1100℃ for a long time without any reaction with strong acid and alkali, and it has the complete light transmittance(pure transparent).
The fluorphlogopite mica has the following unique characteristics:
- highly chemical stability (even in contact with concentrated hydrochloric or sulfuric acid)
- excellent transmittance from UV to IR,
- outgasing-free at high temperature in vacuum,
- excellent electrical insulation,
- high heat endurance (from -100 °C up to 1100 °C),
- non-radioactive background,
- insensitive to the conditions for long-term storage
which are much superior to that of natural mica.
The observation window of water level gauge on high pressure boiler is made of traditional natural mica, which is brown, bad light transmittance and temperature range at 200-700℃, especially its weak corrosion resistance. In thermal power plant, the steam drum water in boiler contains alkali. The natural mica, after the reaction with alkali and the wash by hot water, will be easily pilling, fouling and broken. The result of which is that the water level will not be clear at the short period of time (about 1to 2months), and after being broken, it extremely easy to cause the leakage.
 Fluorphlogopite mica does not react with acid-base solution and has not hydration reaction with water, so it is not layer, not dirty and no rupture. Under the long-term scour by high temperature and high pressure water, the fluorphlogopite mica can still maintain the original transparency and clarity. Fluorphlogopite mica has been widely applied as observation window of steam drum water level gauge of high pressure boiler.
Fluorphlogopite mica does not react with acid-base solution and has not hydration reaction with water, so it is not layer, not dirty and no rupture. Under the long-term scour by high temperature and high pressure water, the fluorphlogopite mica can still maintain the original transparency and clarity. Fluorphlogopite mica has been widely applied as observation window of steam drum water level gauge of high pressure boiler.
The synthetic mica sheets are suitable for a variety of uses such as substrates of thin film, windows and monochromater for x-ray, neutron diffraction, microwave and optics, spacers of electrical vacuum devices, supporters at high temperature and water-gauges of high pressure boilers, and etc., which are important in modern industry and most advanced branches of science. Fluorphlogopite mica plates are used eg. in radar technology, electron microscopes and medical equipment, aerospace and electronic components.
Apart from the use of plates made of pure fluorphlogopite, synthetic mica is used in processed form. The main product is the mica paper obtained from synthetic mica, and it, in turn, is a base for the production of micanite plates, tapes, tubes, parts etc used in temperatures up to 1100 °C.
Our trade offer includes both synthetic mica in the form of plates and discs as well as ground mica of various granulation degree: from powder (grains about 5 μm) to fine flakes (about 0.4 mm). More information on our webpage Synthetic mica.
Synthetic mica properties
Electric Performance
The fluorphlogopite mica, because of its purity, has high body resistivity (1000 times higher than natural mica) and can be safely used at the temperature up to 1100°C. Natural mica, due to its impurity, has unstable electric performance, especially at high temperature and high frequency. When above 500°C, it will gradually lose the characteristics of original because of dehydration.
Performance of Vacuum Deflation
The vacuum deflation of fluorphlogopite mica is low, and only the absorbed gases such as O2 , N2 and Ar are detected by a mass spectrograph. As no vapor of H2O is emitted, it is recommendable as electric vacuum insulant with the vacuum components’ service life greatly prolonged.
The natural mica gives off H2O and other volatiles, hence large vacuum deflation, 2000 times higher than fluorphlogopite mica at 900°C.
Physical Performance
Fluorphlogopite mica is not inclined to distortion and can bear big stress, tension and compression.
Heat-resistance Performance
Fluorphlogopite mica has superior thermal stability. It can be used up to 1100℃ with its thickness almost unchanged, and will decompose gradually at 1200℃. Its melting or crystallization temperature is (1350±5)℃.
Natural mica, as containing (OH) -, tends to be agravic at 200℃, and begins to decompose above 450℃ while thickening, severely agravic above 600°C and almost completely decomposing at 900°C. Therefore, the use of natural mica is restricted. Synthetic mica is therefore recommended for high temperature applications up to 1100 ℃. It can also work in sub-zero temperatures to - 100 ℃.
Optical Performance
Fluorphlogopite mica, because of its small impurity, it has good photopenetrativity from ultraviolet to infrared. Ultraviolet almost cannot penetrate natural mica, but can penetrate fluorphlogopite mica by nearly 0.2 microns. The natural mica has a distinct absorption peak at 2.75 microns, while fluorphlogopite mica has not, which is an important indication that fluorine replaces hydroxyl. So we can conclude that fluorphlogopite mica is a sort of good euphotic material from ultraviolet (0.2 microns) to infrared (4.5 microns).
| Photopenetrativity of fluorphlogopite mica | |
| in UV and visual range | in IR range |
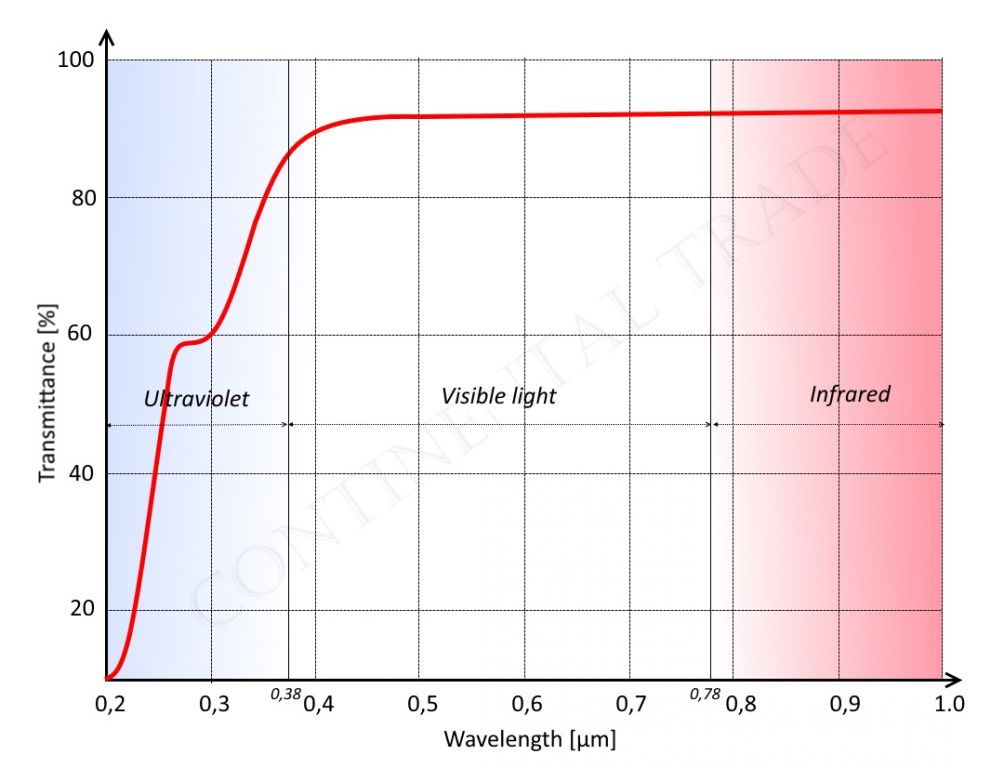 |
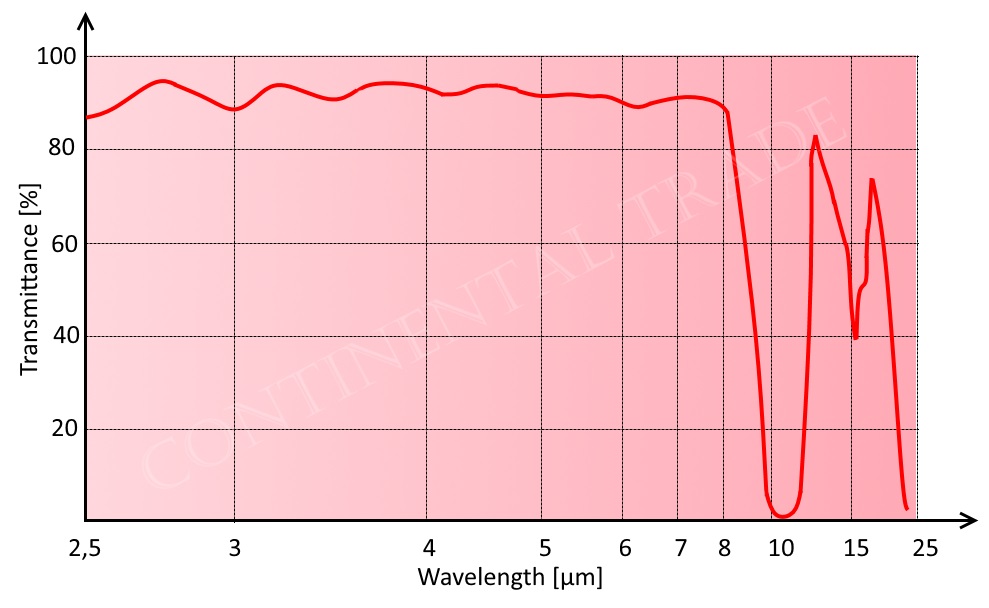 |
Anticorrosion
Fluorphlogopite mica will not react with common acid and alkali solutions, neither will it with water. For that reason, it will not become layered, dirty and disrupted. It will almost remain its original clarity and transparence after being washed by water (either acid or alkali) of high temperature and high pressure for a long time (2-3 years).
Natural mica will become buff and hydrated after being washed by heated and pressurized water or overheated vapor for months. As a result, it becomes much less transparent and layered, dirty, even nontransparent and disrupted. Even long term immersion in hydrochloric or sulfuric acid is not a problem.
| PROPERTIES | ||
|---|---|---|
| Type | Units | Parameter |
| Long run work temperature | °C | from -100 up to 1100 |
| Melting temperature | °C | 1350 ±5 |
| The heat loss (40°C - 1180°C ) | % | 0.85 |
| Water absorption rate | % | 0.14 - 0.23 |
| Moisture absorption rate | % | 0 - 0.05 |
| Specific heatc apacity | J / kg .K | 836 |
| Coefficient of linear expansion | 1/K | (‖)10 - 12 × 10-6 (⊥)15 - 25 × 10-6 |
| Thermal conductivity | W/m·K | (‖)3.75 (⊥)0.55 |
| Density | g/cm3 | 2.78 - 2.85 |
| Dielectric strength | kV/mm | 185 -238 |
| Dielectric constant (ε) | 5.6 | |
| Dielectric loss (tg δ) @ 1 Mhz | (1 - 4) · 10-4 | |
| Volume resistivity | Ω . cm | 1015 - 1016 |
| High temperature mica volume resistivity (@ 300°C; 500°C; 600°C; 700°C; 800°C) |
Ω . cm | 3.9×1015; 5.1×1013; 1.6×1012; 5.6×1011; 3.9×1010 |
| Surface resistivity | Ω . cm | 1011 - 1013 |
| Tensile strength | MPa | 150 - 200 |
| Compressive strength | MPa | 400 - 520 |
| Elastic modulus | kg/mm2 | 17 500 |
| 3-point bending strength | ||
| - at room temperature | MPa | 45.6 |
| - at temperature 200°C | MPa | 29,8 |
| Optical angle | ° | 8 - 14 |
| Refractive index | np1.513 - 1.544; nm1.539 -1.564; ng1.540 - 1.566 | |
| Hardness | Mohs | 3 - 3.4 |
| icro hardnessHv0.2 | 1.35 Gpa; S 138 to 146 | |
| Mosaic of (Mosaicity ) | <0.25 | |
| Cleavage plane(@200°C;600°C;900°C) | 10; 6.3; 2.96 | |
| Cleavage surface (@200°C;600°C;900°C) | 21.2; 11.5; 5.08 | |
Comparison between synthetic and natural mica
| Synthetic mica | Natural mica | |
|---|---|---|
| Quality consistency |
Synthetic mica is synthesized by internal heat. All the raw materials for each batch and the formula can be controlled, which guarantees the quality consistency of each batch. | Due to the mine is formed for millions of years, the quality of each mica mine is quite different. Even though in a same mine, the quality is hard to keep consistency. Thus, the quality of each batch is different. |
| Radiation | Synthetic mica is synthesized artificially so that it is free from natural radiation. | Most of ores are more or less with radiation as the mine was formed through millions of years. |
| Purity | Synthetic mica is a crystallized combination produced according to formula. The raw materials are by severe screening, which guarantees the high purity. | The mica is formed by natural powder so that it is with many mineral impurities. Meanwhile, it gets pollution during mining. The purity is much less than synthetic mica. |
| Heavy metal content | Synthetic mica is nearly free from heavy metal. | Natural mica is with many harmful heavy metal to human health. |
| Appearance | Smooth surface. High whiteness(>92). | It has worse surface smoothness and looks grey. The whiteness is about 60 - 80. |
| Heat-resistance | The melting point of synthetic mica reaches(1350±5)℃. | Generally at 200 ℃, the layers start to stratify and the color changed. |
| Electric property | Dielectric strength: 185-238 kV/mm | Dielectric strength: 115-140 kV/mm |
| Insulation property | Surface resistance: 3×1013 Ω | Surface resistance: 1×1011 Ω |
| Tensile strength | 150 Mpa | 110 - 140 Mpa |
| (OH)Bond | Synthetic mica is OH Bond free. It is not like the natural mica would release harmful hydrogen to steels. Thus, it can be used to antirust metal products. | |
| Optically transparent rate | Synthetic mica crystal is colorless and transparent that has better optically transparent property against infrared and ultraviolet radiation than natural mica does. So the synthetic mica based pearl pigment is quality. |
Comparison of some parameters
| Unit | Synthetic mica | Phlogopite | Muscovite | |
|---|---|---|---|---|
| Density | g/cm3 | 2,8 | 2,6 - 3,2 | 2,6 - 3,2 |
| Dielectric strength | kV/mm | ~ 180 | 115 - 140 | 120 - 200 |
| Volume resistivity | Ω.cm | (3-6)×1015 | 1012-1014 | 1013-1017 |
| Surface resistivity | Ω | 3×1013 | 1010-1014 | 1011-1012 |
| Dielectric constant | ε | ~ 6 | 5,0 - 6,0 | 6,0 - 7,0 |
| Dielectric loss | tg δ | 3 × 10-4 | (10-50)×10-4 | (1- 4)×10-4 |
| Tensile strength | kg/cm2 | ~1500 | ~ 1000 | ~ 1750 |
| Water absorption | % | 0,14 | 2,7 | 2,2 |
| Moisture absorption rate | % | 0,04 | 0,24 | 0,18 |
| Heat resistance | ℃ | > 1100 | 600 - 650 | 350 - 450 |






